Exploring the Relationship Between Angiogenesis Inhibition of sFLT-1 and Sickle Cell Nephropathy
Main Article Content
Keywords
Sickle Cell Disease, sFLT-1, angiogenesis, renal damage, VEGF, microalbuminuria, endothelial dysfunction
Abstract
Background: Sickle cell anaemia (SCA) is a chronic haemoglobinopathy associated with multi-organ complications, among which nephropathy remains a significant cause of morbidity and mortality. Early detection of sickle cell nephropathy (SCN) is crucial, yet conventional markers such as serum creatinine and estimated glomerular filtration rate (eGFR) often fail to detect early renal impairment. Emerging biomarkers like soluble fms-like tyrosine kinase-1 (sFLT-1) have shown potential in identifying early endothelial and glomerular injury in SCA. This study explores the role of angiogenesis inhibition via sFLT-1 in the pathogenesis of renal injury in SCD, aiming to identify correlations between elevated sFLT-1 levels, proteinuria, and declining renal function.
Methodology: A cross-sectional analytical study was conducted involving confirmed SCD patients and age- and sex-matched controls. Serum sFLT-1 levels were measured using enzyme-linked immunosorbent assay (ELISA). At the same time, renal function was assessed through estimated glomerular filtration rate (eGFR), serum creatinine, and urinary microalbumin and microalbumin: creatinine ratio quantification. Statistical analyses included correlation studies and group comparisons to determine the association between sFLT-1 concentrations and renal impairment indicators.
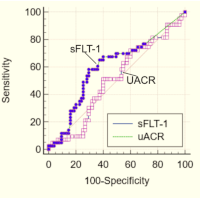
Results: SCD patients demonstrated significantly elevated sFLT-1 levels compared to controls (p < 0.001). Higher sFLT-1 levels were strongly associated with increased albuminuria and reduced eGFR, indicating early renal damage. These findings suggest that elevated sFLT-1 contributes to glomerular endothelial dysfunction, possibly via inhibition of vascular endothelial growth factor (VEGF)-mediated repair mechanisms.
Conclusion: The study reveals a significant relationship between angiogenesis inhibition by sFLT-1 and renal dysfunction in individuals with SCD. sFLT-1 may serve as a promising early biomarker for SCD nephropathy and a potential therapeutic target to preserve renal function in this vulnerable population. Furthermulticentred and longitudinal studies are recommended to establish causality and explore interventional strategies.
References
2. Brewin J, Tewari S, Hannemann A, Balushi H Al, Sharpe C, Gibson JS, et al. Early Markers of Sickle Nephropathy in Children With Sickle Cell Anemia Are Associated With Red Cell Cation Transport Activity. Hemasph Eur Hematol 2017;1(1):1–7.
3. Safdar, Osama & Baghdadi, Rana & Alahmadi, Sereen & Fakieh, Bana & Algaydi, Amaal. (2022). Sickle cell nephropathy: A review of novel biomarkers and their potential roles in early detection of renal involvement. World Journal of Clinical Pediatrics. 11. 14-26. 10.5409/wjcp.v11.i1.14.
4. Flavia C. M. Lopes, Fabiola Traina, Camila B. Almeida, Flavia C. Leonardo, Carla F. Franco-Penteado, Vanessa T. Garrido, Marina P. Colella, Raquel Soares, Sara T. Olalla-Saad, Fernando F. Costa, Nicola Conran. Key endothelial cell angiogenic mechanisms are stimulated by the circulating milieu in sickle cell disease and attenuated by hydroxyurea. Haematologica 2015;100(6):730-739; https://doi.org/10.3324/haematol.2014.119727.
5. Hoon Chang, Shanshan Gustafson and Tulsi Mehta. The Vascular Endothelial Growth Factor (VEGF) Pathway Inhibition and Associated Nephrotoxicities. Chapter 8, Chronic Kidney Disease.IntechOpen.2022. doi: 10.5772/intechopen.1000212, https://doi.org/ 10.5772/intechopen.1000212.
6. Matsui M, Onoue K, Saito Y. sFlt-1 in Chronic Kidney Disease : Friend or Foe ? Int J Mol Sci. 2022;23:1–13.
7. Failla CM, Carbo M, Morea V. Positive and negative regulation of angiogenesis by soluble vascular endothelial growth factor receptor-1. Int J Mol Sci. 2018;19(5).
8. Ataga, Kenneth I., Derebail, Vimal K, Archer, David R. The glomerulopathy of sickle cell disease. Am. J. Hematol..89(9).. 2014. https://doi.org/10.1002/ajh.2376 https://doi.org/10.1002/ajh.23762. 907- 914.
9. Doi K, Noiri E, Fujita T. Role of vascular endothelial growth factor in kidney disease. Curr Vasc Pharmacol. 2010;8(1):122–128.
10. Kato GJ, Steinberg MH, Gladwin MT. Intravascular hemolysis and the pathophysiology of sickle cell disease. J Clin Invest. 2017 Mar 1;127(3):750-760. doi: 10.1172/JCI89741. Epub 2017 Mar 1. PMID: 28248201; PMCID: PMC5330745.
11. Schulz A, Drost CC, Hesse B, Beul K, Brand M, Di Marco GS. The Soluble Fms-like Tyrosine Kinase-1 Contributes to Structural and Functional Changes in Endothelial Cells in Chronic Kidney Disease. Int J Mol Sci. 2022 Dec 16;23(24):16059. doi: 10.3390/ijms232416059. PMID: 36555698; PMCID: PMC9787493.
12. Vishwakarma G. Sample Size and Power Calculation. In: Statistical Basis of Calculation of Sample Size in Nursing Research. Nur Res 2020.235–246.
13. Ballas S. More definitions in sickle cell disease: Steady state v base line data. Am J Hematol. 2012;87:338-347.
14. Poudel B., Yadav B.K., Nepal A.K., Jha B., Raut K.B. Prevalence and association of microalbuminuria in essential hypertensive patients. North Am. J. Med. Sci. 2012;4:331–335. doi: 10.4103/1947-2714.99501. [DOI] [PMC free article] [PubMed] [Google Scholar]
15. Manu M, Akuyam SA, Aminu SA, Hassan A, Adamu R, Suleiman HM, et al. Assessment of some biochemical markers of nutritional Status among adult sickle cell anemia patients in Steady state in north-west Nigeria. Journal Abu Edu Ng 2019;18(5):459–64.
16. Asafa MA, Ahmed IO, Ogunlade O, Bolarinwa RA. Arterial blood pressure indices among young adults with sickle cell anaemia in Nigeria. J Cardiol Curr Res. 2023;16(5):122–126.
17. Ataga KI, Brittain JE, Moore D, Jones SK, Hulkower B, Strayhorn D, et al. Urinary albumin excretion is associated with pulmonary hypertension in sickle cell disease: Potential role of soluble fms-like tyrosine kinase-1. Eur J Haematol. 2010;85(3):257–263.
18. Laurentino MR, Parente Filho SLA, Parente LLC, da Silva Júnior GB, Daher EDF, Lemes RPG. Non-invasive urinary biomarkers of renal function in sickle cell disease: an overview. Ann Hematol. 2019 Dec;98(12):2653–2660. .
19. Niss O, Lane A, Asnani MR, Yee ME, Raj A, Creary S, et al. Progression of albuminuria in patients with sickle cell anemia: A multicenter, longitudinal study. Blood Adv. 2020;4(7):1501–1511.
20. Youssry I, Makar S, Fawzy R, Wilson M, AbdAllah G, Fathy E. Novel marker for the detection of sickle cell nephropathy: soluble FMS-like tyrosine kinase-1 (sFLT-1). Pediatr Nephrol. 2015; 30(12):2163–2168.
21. Bolarinwa RA, Akinlade KS, Kuti MA, Olawale OO, Akinola NO. Renal disease in adult Nigerians with sickle cell anemia: a report of prevalence, clinical features and risk factors. Saudi J Kidney Dis Transpl. 2012;23(1):171–175.
22. Ranque B, Menet A, Diop IB, Thiam MM, Diallo D, Diop S, et al. Articles Early renal damage in patients with sickle cell disease in sub-Saharan Africa : a multinational, prospective, cross-sectional study. Lancet Haematol. 2014;e64-73.
23. Vazquez B, Shah B, Zhang X, Lash JP, Gordeuk VR, Saraf SL. Hyperfiltration is associated with the development of microalbuminuria in patients with sickle cell anemia. Am J Hematol. 2014;89(12):1156–1157.
24. Gbotosho OT, Kapetanaki MG, Kato GJ. The Worst Things in Life are Free: The Role of Free Heme in Sickle Cell Disease. Front Immunol. 2020;11:561917.
25. Payán-Pernía S, Ruiz Llobet A, Remacha Sevilla ÁF, Egido J, Ballarín Castán JA, Moreno JA. Sickle cell nephropathy. Clinical manifestations and new mechanisms involved in kidney injury. Nefrol. 2021;41(4):373–382.
26. Zahr RS, Saraf SL. Sickle Cell Disease and CKD: An Update. Am J Nephrol. 2024;55(1):56–71.
27. Akuyam SA, Abubakar A, Lawal N, Yusuf R, Aminu SM, Hassan A, et al. Assessment of biochemical liver function tests in relation to age among steady state sickle cell anemia patients. Niger J Clin Pract. 2017;20(11):1428–1433.
28. Payán-Pernía S, Ruiz Llobet A, Remacha Sevilla ÁF, Egido J, Ballarín Castán JA, Moreno JA. Sickle cell nephropathy. Clinical manifestations and new mechanisms involved in kidney injury. Nefrologia. 2021;41(4):373–382.
29. Nnaji UM, Ogoke CC, Okafor HU, Achigbu KI. Sickle Cell Nephropathy and Associated Factors among Asymptomatic Children with Sickle Cell Anaemia. Hindawi Int J Pediatr. 2020;2020:1–8.
30. Payán-Pernía S, Ruiz Llobet A, Remacha Sevilla ÁF, Egido J, Ballarín Castán JA, Moreno JA. Sickle cell nephropathy. Clinical manifestations and new mechanisms involved in kidney injury. Nefrologia. 2021;41(4):373–382.
31. Gounden V, Bhatt H JI. Renal Function Tests. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. 2024. p. 221-230
32. Waikar SS, Bonventre J V. Creatinine kinetics and the definition of acute kidney injury. J Am Soc Nephrol. 2009: 20(3):672–679.
33. Ataga KI, Brittain JE, Jones SK, May R, Delaney J, Strayhorn D, Desai P, Redding-Lallinger R, Key NS, Orringer EP. Association of soluble fms-like tyrosine kinase-1 with pulmonary hypertension and haemolysis in sickle cell disease. Br J Haematol. 2011 Feb;152(4):485-91. doi: 10.1111/j.1365-2141.2010.08410.x. Epub 2011 Jan 11. PMID: 21223248.
34. Ataga KI, Saraf SL, Derebail VK. The nephropathy of sickle cell trait and sickle cell disease. Nat Rev Nephrol. 2022 Jun;18(6):361-377. doi: 10.1038/s41581-022-00540-9. Epub 2022 Feb 21. PMID: 35190716; PMCID: PMC9832386.
35. Landburg PP, Elsenga H, Schnog JB, Duits AJ. Increased serum levels of anti-angiogenic factors, soluble fms-like tyrosine kinase and soluble endoglin in sickle cell disease. Acta Haematol. 2009;120(3):130–123.
36. Rajakumar A, Michael HM, Rajakumar PA, Shibata E, Hubel CA, Karumanchi SA, et al. Extra-placental expression of vascular endothelial growth factor receptor-1 (Flt-1) and soluble Flt-1 (sFlt-1), by peripheral blood mononuclear cells (PBMCs) in normotensive and preeclamptic pregnant women. Placenta. 2005;26(7):563–573.